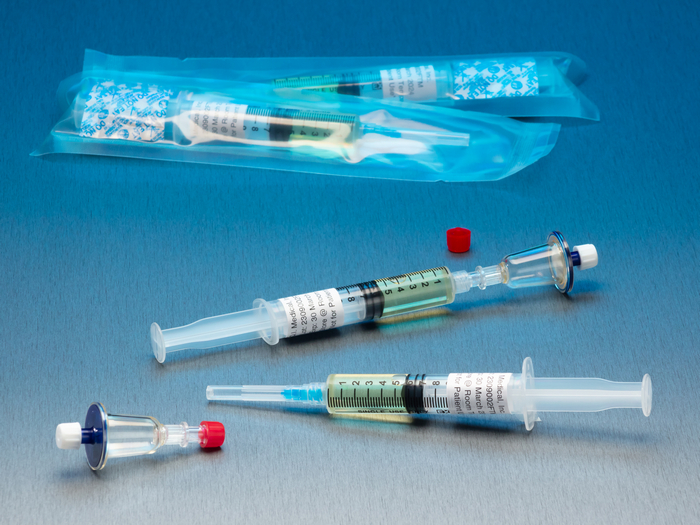
QT Micro™ System – TM6001
10 units / case
Overview
Full filtration microbial contamination testing system for small volume compounded sterile products.
Nondestructive test allows for sample to be used after testing.
FDA Registered Class 2 Device with required FDA Unique Device Identifier (UDI) barcode.
0.22µ microbial contamination test for syringe to syringe transfers. If you are using gravity or vacuum assisted filtration, see our QT Junior™ System TJ3100.
- 10 x QT Micro filter units with female luer lock inlet and female luer lock outlet. For filter units with female luer lock inlet and male luer slip outlet, see QT Micro™ System – TM6101.
- 10 x 5cc syringes of FTM growth media
- Log sheet included
- Gummed labels included
Certificate of Analysis Search
Enter the Lot Number below for a PDF file of the COAs: